
When comparing my experiences with those who came before me, I’m happy to realize that cancer treatments have gotten better and there are more Black leaders in the medical community.


When comparing my experiences with those who came before me, I’m happy to realize that cancer treatments have gotten better and there are more Black leaders in the medical community.

Women who face menopause after a cancer diagnosis may be able to manage their symptoms without relying on estrogen-based therapies, although discussing this with a cancer team will help patients find what’s best for them.

I knew my family was close, but it wasn’t until my mom was diagnosed that I realized how bonded we truly were.
Vaginal cancer is amongst the rarest of gynecologic cancer, it is important those patients with a rare cancer advocate for themselves and find doctors who are experts in treating it.

From Blink-182’s bassist Mark Hoppus discussing his cancer experience in an upcoming book to Bruce Sutter, a Hall-of-Fame pitcher and Cy Young winner, dying of the disease, here’s what’s happening in the cancer space this week.
NC410 plus Keytruda will be studied in a phase 1b/2 trial for patients with certain solid cancers that either did not respond to or have not been treated with an immunotherapy agent.

The truth is I stagger back and forth between the two camps — tell it like it is or walking on sunshine. It depends on the day, my treatment regimen, the lab or scan results, the losses and the gains.

My healthy body was hijacked by an enemy (ovarian cancer) launching my continuous fight against the disease.

From “Chicago Med” actress Marlyne Barrett announcing her gynecologic cancer diagnosis to a New York City-based news team walking in honor of their former news director who died of glioblastoma, a form of brain and spinal cord cancer, here’s what’s happening in the cancer space this week.

The FDA’s approval of Vegzelma was based on data that demonstrated the biosimilar elicited comparable safety and efficacy to Avastin in several studies.
The OnPrime trial will investigate if adding Olvi-Vec, a modified virus, to chemotherapy and Avastin will improve outcomes for pretreated platinum-resistant or refractory ovarian cancer.

Maintenance therapy helps deter undetectable cancer that may exist after initial treatment.
Treatment with Libtayo may improve survival over a 30-month period with no new side effects in patients with recurrent or metastatic cervical cancer, opening the doors for an immunotherapy option for these patients.
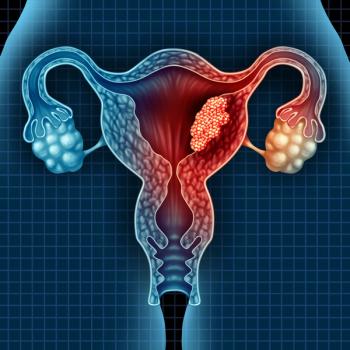
Lenvima plus Keytruda improved survival outcomes for patients with pretreated advanced endometrial cancer, according to recent clinical trial data.
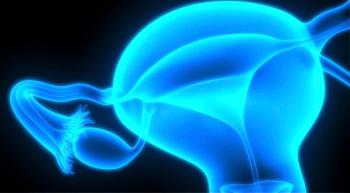
Rubraca improved progression-free survival in patients with BRCA1/2-mutant, platinum-sensitive, relapsed ovarian cancer, according to findings from the ARIEL4 clinical trial.

Cervical cancer survivor Ginny Marable heard about a story of a fellow cervical cancer survivor, Tamika Felder, not being able to preserve her fertility. The story moved Marable so much that she and her husband donated their embryos to Felder.

I am still here fighting, fighting hard and I will continue to do so to make sure I have more graduations, more weddings, and more everything that I can squeeze into my precious time left here on earth.
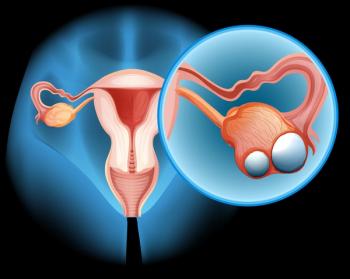
Adding Rivoceranib to pegylated liposomal doxorubicin — a type of chemotherapy — improved survival outcomes for patients with platinum-resistant ovarian cancer, according to recent study results.

The manufacturer of one of the cancer treatments announced that it is also participating in clinical trials assessing the safety and efficacy of their product in colorectal and endometrial cancer.

The study the FDA allowed to continue is investigating the safety and efficacy of the novel cancer drug ACR-368 in patients with endometrial, urothelial and ovarian cancer.

Tafinlar plus Mekinist is now approved by the Food and Drug Administration to treat adults and children over the age of 6 who have BRAF V600-mutant advanced solid cancers.

The FDA approval of the first poly (adenosine diphosphate-ribose) polymerase, or PARP, inhibitor in the 2010s has drastically changed how patients with gynecologic cancers are treated.

The use of newer tools and techniques may be reducing the risk of surgical site infections and other complications in patients receiving treatment for gynecologic cancers.

I was told that I would miscarry my pregnancy, but continued to feel more pregnant, leading to my molar pregnancy diagnosis — a condition I’ve never heard of before.

The Strata PATH trial will use a novel testing strategy to determine if certain patients with solid tumors can benefit from already-approved therapies.